Formerly Blind Contestant, Christian Guardino, Gets Golden Buzzer on AGT

Sixteen-year-old singer Christian Guardino is riding high this week. He got a coveted Golden Buzzer on America’s Got Talent, advancing him directly to the live shows.
But when you realize that Christian was cured of a previously incurable form of blindness, his story becomes even more remarkable.
Shortly after birth, Christian’s mother, Elizabeth, knew something was not right with her son. His eyes jiggled and jerked and he only seemed to focus on very bright lights. An eye doctor referred them to New York’s Mount Sinai Hospital, where Christian underwent a test called an electroretinogram (ERG). Electroretinography is an eye test used to detect abnormal function of the retina (the light-detecting portion of the eye). Specifically, in this test, the light-sensitive cells of the eye, the rods and cones, and their connecting ganglion cells in the retina are examined. Christian’s test showed very poor function of the retina, and he was diagnosed with Leber congenital amaurosis.
The doctors told Elizabeth that Christian’s eyesight was very poor and would remain so, as there were no treatments for the condition. Christian would have to learn to walk with a cane.
In 2012, at age 12, the Guardinos visited a clinic run by the University of Pennsylvania’s Scheie Eye Institute. There he was enrolled in a clinical trial with the team of Dr. Jean Bennett. Dr. Bennett had determined that Christian had a specific defective gene, known as RPE65, that she hope to insert into a virus and then inject into his eyes to repair the defect.
It worked! Three days after surgery, in a dimly lit room, Christian looked over at the woman at his bedside and said, “Mom, is that you?” The procedure restored his sight to between 20/50 and 20/70. He no longer needs a cane, and can read enlarged notes on sheet music.
Good luck Christian, we’re rooting for you!
What is Leber congenital amaurosis?
 Leber congenital amaurosis (LCA) is an eye disorder that primarily affects the retina, which is the specialized tissue at the back of the eye that detects light and color. People with this disorder typically have severe visual impairment beginning in infancy. The visual impairment tends to be stable, although it may worsen very slowly over time.
Leber congenital amaurosis (LCA) is an eye disorder that primarily affects the retina, which is the specialized tissue at the back of the eye that detects light and color. People with this disorder typically have severe visual impairment beginning in infancy. The visual impairment tends to be stable, although it may worsen very slowly over time.
Leber congenital amaurosis is also associated with other vision problems, including an increased sensitivity to light (photophobia), involuntary movements of the eyes (nystagmus), and extreme farsightedness (
A specific behavior called Franceschetti’s oculo-digital sign is characteristic of Leber congenital amaurosis. This sign consists of poking, pressing, and rubbing the eyes with a knuckle or finger.
At least 13 types of Leber congenital amaurosis have been described. The types are distinguished by their genetic cause, patterns of vision loss, and related eye abnormalities.
Leber congenital amaurosis occurs in 2 to 3 per 100,000 newborns. It is one of the most common causes of blindness in children.
How is LCA treated?
Except for RPE65-related LCA, no substantial treatment or cure for LCA exists, and, thus, care is supportive. Parents should be referred to programs for the visually impaired child within their state or locality.
Affected individuals benefit from correction of refractive error, use of low-vision aids when possible, and optimal access to educational and work-related opportunities.
Gene Therapy for RPE65-related LCA
As mentioned above, there are at least 13 types of LCA, each caused by different mutations in the genes responsible for vision. In the type of LCA that Christian has, there is a mutation in a gene called RPE65. The protein made by this gene is a crucial component of the visual cycle. The RPE65 protein is necessary for the production of a retina-specific form of vitamin A that is required for the light-sensitive photoreceptor cells to function. Mutations in the RPE65 gene prevent this production, which halts the visual cycle and blocks vision.
Researchers at the University of Pennsylvania, Philadelphia, and the University of Florida, Gainesville were able to isolate the gene and insert its DNA into a benign virus. Unfortunately, many other genes that lead to LCA are too big to fit into the viral DNA, making this method not possible for patients with those mutations. The modified virus is grown in culture and subjected to a complex concentration and purification procedure.
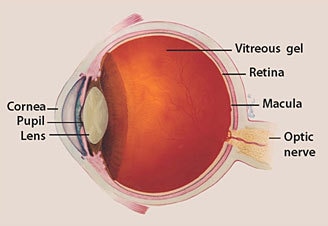
During surgery, the doctor removes a portion of the vitreous gel (see diagram above) closest to the retina. A solution containing the modified virus is then injected into that space. The hope is that the virus would “infect” the retinal cells, thereby bringing a normal copy of the diseased gene to the affected tissues.
Although there have not been many patients treated with this procedure, the results have been hopeful- especially as seen in Christian’s case.



























0 comments